Kitmondo
29 May 2014
This article was originally published in April 1907 by Peter Evermann. Information within this article is therefore correct as of 1907. The publication of this material aims to provide historical insight on the subject and its place in industry.
Open-hearth steel versus Bessemer steel—this is the great struggle between two rivals in modern compound steel manufacturing plants. The average user of open-hearth steel claims that this quality of steel is better than the Bessemer. The manufacturer, however, has much less trouble and costs with the latter. Therefore, it is his intention to keep the market up with Bessemer quality.
The complaint has been frequently heard that for many applications this material is less suited than that produced in the open-hearth. From a scientific standpoint it is difficult to see why the latter should be superior, since the chemical end result in both can be made entirely alike. But there is a physical explanation for this practical inequality. The blowing operation is probably too rapid to permit keeping pace with the reactions within the modern large sizes of Bessemer converters all uniformly through the entire bath of molten metal; there is also no time for thorough continuous chemical analysis. And even when the spectroscope is used for observation of the flame, one merely depends on the physical tests and senses of the assayer or men in charge.
Of course, since the general adoption of the mixer, the blowing process, for both acid and basic converters, was so far simplified that any question of a difference between Bessemer and Martin quality could have been entirely disregarded, because the composition of the charged liquid pig iron which is drawn off from the mixer is definitely determined beforehand. But the old prejudice exists yet.
In the open-hearth process, however, the mixer never played such an important part, because there was always ample time for both chemical and physical tests and observations, and the uniform quality was thus assured without resorting to the use of the expensive mixer. But also in this case it is much easier to produce the desired quality of steel by using the mixer, as the pig iron contains always an approximately uniform and even percentage of silicon, carbon, manganese and phosphorus.
At the present time, therefore, in the various methods of steel production the governing factor has come to be not so much a question of quality as one of costs and quantity. The Bessemer process is still unrivaled as regards its cheapness, and the Siemens-Martin process still unrivaled concerning quality and cheapness, the latter only in comparison with the expensive crucible steel. The average production of an open-hearth furnace is yet about 100 tons in 24 hours and 800 tons daily for the Bessemer furnace. The output of Thomas converters is generally less. It may, therefore, be safely assumed that one open-hearth is equivalent to only one-seventh of a converter in point of production, which fact affords an excellent indication of the greater capacity of the converter steel works of to-day.
Blast Furnace Gas
Now, since human knowledge of iron ore started, since those gray historical times up to date, the idea was never dropped to make steel straight from iron ore. The readers of this journal are, of course, acquainted with this history, and it is not the purpose of these lines to repeat known things. I will mention here only the recent developments of various electrical methods—in my mind all ingenious theoretical experiments, but impractical yet.
Before going into the real subject I may call attention to the recent development of the modern gas engines driven by blast furnace gas. For many years this gas was escaping unseen in daytime, and the bright light from the furnace tops was throwing its shine into the night down on the human buildings, like light fires are guiding the ships on the ocean now. In this way uncounted millions have been and are still being lost. It is only a few years ago those even up-to-date engineers asked the question, "What has a gas engine got to do with a blast furnace?" But now so much has already been written on this subject that I will note only the main points to be considered in this question.
The blast furnace of the future will be considered as a big gas producer. The gas might be used in gas engines to generate electricity for general power purposes, or it may be used in gas furnaces for the manufacture of steel or other products. There is already also some talk of the entire abandonment of the coke ovens. But as this field is not yet so far developed, I have to start with those fuel furnaces for the blast furnaces.
The following calculation is of minor influence on my com-pound-steel furnace, but it is a part of it, as it proves that this coke oven gas is available in sufficient quantities in modern by-product plants so as to allow its further use in steel furnaces or in gas engines. Eleven thousand tons of soft coal yield in average about 10,000 tons of dry coal, and are converted to about 7,850 tons of coke.
Practice has proved that 1 ton of coal is equivalent to about 9,000 cubic feet or even more of such gas. The heating value is somewhat lower than that of city gas, averaging, say, 500 B.T.U. per cubic foot. A good gas engine will yield 1 hp. per hour for 10,000 B.T.U. But considering less favorable conditions, 12,000 B.T.U. may be assumed in this calculation; 24 cubic feet per hour of this gas are therefore required per horse-power.
Fifty per cent of all gas has to be taken for heating the coke ovens, and if ten further per cent are deducted for uncontrollable leakage, only 3,600 cubic feet of gas are available for power per ton of coal, or 3,600 ÷ 24 = 150-hp. hours for each ton of coal charged in the coke oven.
This result can be checked in the following way: One pound of coke oven gas is equivalent to about 33 cubic feet at normal temperature; 2,000 pounds, or 1 ton of coke gas measures 66,000 cubic feet, hence the above noted 1,600 tons of coke oven gas measures 105,600,000 cubic feet of gas. Sixty per cent off for heating and loss leaves for power 42,240,000 cubic feet, yielding 1,760,000-hp. hours. The quantity of coal charged was 11,000 tons, and 1,760,000 ÷ 11,000 gives 160-hp. hours per ton of coal charged.
Practical experience shows that in a blast furnace plant there are required about 8 to 10 hp. per ton of pig iron produced to run the plant. A 500-ton furnace would require, therefore, about 5,000-hp. machinery. It has already been claimed that a coke plant near the blast furnace plant is sufficient to run the latter. Is it true?
Five hundred tons of pig will require, roughly, 500 tons 01 coke; 1 ton coke equals 1.3 to 1.4 tons of coal. If we consider only 1.3 tons, we have 500 X 1.3 = 650 tons of coal charged in the coke ovens in 24 hours. Hence, 650 X 150 = 97,500-hp. hours will be available from the coke plant during 24 hours. This means that there are continually available 97,500 ÷ 24, or approximately, 4,000 hp. If we assume a power requirement of only 8 hp. for the ton of pig iron, these 4,000 hp. cover exactly the requirements of the 500-ton furnace plant.
If I can cover, -therefore, the wants of my blast furnace plant by my coke ovens, why not try to cover the requirements of my steel plant by the blast furnace plant? Is it possible?
Much has been published in recent years on the proposition to use the available blast furnace gas in gas engines, and many practical results have been obtained in Europe, and more recently also in America. I will, therefore, deal with this subject only in so far as is necessary to find the amount of gas from the blast furnace which is used in the steel plant and will discuss how it is used in a plant.
A considerable number of furnaces are operating now with an average output of 24 tons of pig iron per hour, or 576 tons per day of 24 hours. For convenience in calculation we will use this figure.
With the object of obtaining as much gas as possible from the blast furnace, using it as a large gas producer, the pig iron can be obtained, on paper, at least, as a by-product in modern steel manufacture.
With the object of obtaining somewhat richer blast furnace gas than may be obtained by using coke alone, some soft coal may be charged into the furnace with the coke. It may be preferable under certain conditions to mix coke oven gas with the blast furnace gas in order to secure a more uniform gas or to install a "mixer" also for all gases of one complete plant.
For each ton of pig iron we get 150,000 cubic feet of gas, having an average heating value of 100 B. T. U. per cubic-foot. If, therefore, all gas would be transformed into power; then 150,000 X 100 = 15,000,000 B.T.U. would be at disposition for each ton of iron. The gas engine requires 12,000 B.T.U. per hour to produce 1 hp., so that 1 ton of iron per hour will yield 1,250 hp., or in a 576-ton daily plant about 30,000 hp. will be available. In the near future a good deal of the heat which is required to heat the blast may be obtained from the liquid cinder, thus doing away with the high and costly hot-blast stoves, but at present we must still consider them as follows:
Twenty-five per cent of the gas of the blast furnaces is to be deducted for heating the stoves. The plant producing 576 tons of pig iron daily, yields 3,600,000 cubic feet of gas per hour; 25 per cent of this amount, or 900,000 cubic feet, are to be set aside for heating the blast.
To supply the required amount of air in the blast furnace the blowing engines are built on an average of, say, 80 to 100 cubic feet of air per pound of coke charged in furnace. For 24 tons per hour are therefore required 2,000 X 24 X 100 = 4,800,000 cubic feet of air per hour, or 80,000 cubic feet per minute. With a pressure of up to 25 pounds per square inch, each 100 cubic feet per minute require about 4 to 5 hp. per hour; hence a furnace of 24 tons of iron per hour requires for its blowing engines from 3,200 to 4,000 hp. The modern gas blower requires 12,000 B. T. U. per horse-power-hour, or if we assume again 100 B.T.U. per cubic foot of gas, about 120 cubic feet of gas. This would result in a further deduction of 480,000 cubic feet of gas for blowing engines, but as these engines are not always running under full power, a figure of 400,000 cubic feet per hour may safely be assumed.
The uncontrollable leakage in an up-to-date plant should be not more than 5 per cent, or 3,600,000 X 0.05 = 180,000 cubic feet per hour.
Mechanically-operated gas-cleaning plants have given better results than plain washing towers; for this purpose are required from 0.4 hp. to 0.7 hp. per 1,000 cubic feet gas. If all gas is cleaned (a proposition which has to be considered in a modern plant) we get 3,600 X 0.7 = 2,520 hp., or 302,400 cubic feet of gas. Since this plant is not always running under full load, 252,000 cubic feet per hour will be a fair figure for this purpose.
For auxiliary machinery, such as ore, lime and coke-hand ling apparatus, electric light plant, power transmission, compressed air, oil and water pumps, elevators and hoists, pig crushers, casting machine, etc., there are required per ton of pig iron per day not more than 2 hp. per hour, or for our 576-ton daily plant a 1,152-hp. engine. Making again some allowance for times of light load, 115,200 cubic feet of gas will be required per hour for this purpose.
Considering 100-B.T.U. quality, this means 175,280,000 B.T.U. per hour for further use in gas engines, which for 12,000 B.T.U. per horse-power-hour means 14,600 hp., or, in accordance with standard gas engine experience, about a 10,000-kw. Plant.
From this we can deduce that the blast furnace as a gas producer covers the costs of manufacturing pig iron. For if we assume an exceptionally high cost for manufacturing pigs at $12.00 per ton, we' have in our 576-ton daily plant an expense of $288 per hour. The 10,000 kw., however, we have in surplus, and can sell them, say, for .22 cents or 3 cents per kw-hour to the public in form of electrical energy. This means an income of $250 to $300 per hour, thus equalizing the entire manufacturing costs.
Steel Manufacturing
From this departure I now return to the steel subject. The main object of my proposition is a new method of manufacturing steel, and this method has the following advantages: (1) The cost of production is reduced; (2) at the same time the tonnage of the output is increased; (3) a less costly plant is required; (4) the plant is independent of the market prices of scrap; (5) less coal is used for the open-hearth quality than hitherto. The writer was very much pleased to find some time ago a similar furnace proposition from another source, working in a large Southern steel plant simultaneously on the same object. An article on this matter was published in Stahl und Eisen, in Germany, by Mr. Goldstein, of Monterey, Mex. I mention this here only to prove that my own standpoint is also based on good practical experience of others.
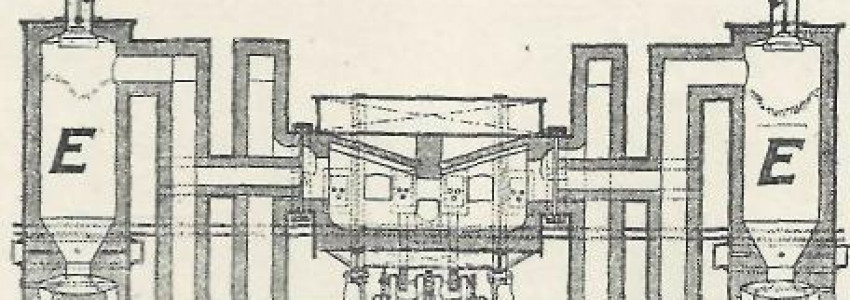
Fig. I shows the general arrangement of my improved steel plant directly attached and operating simultaneously with the blast furnace plant. The blast furnace A may be of any suitable type and construction. My improved type with the all-over-bosh-and-bottom-water-cooling system is shown, however. Like the skyscrapers of to-day, the entire structure rests on a grate of heavy beams a. The charging hoist is shown and indicated by B. The hot blast is supplied by line b, and the blast nozzles shown, too. Slag notch is at c and metal tap at d.
There is no mixer required, as in my mind this expensive vessel is in fact only an unwieldy adjunct, the use of which was rendered compulsory solely on account of the inefficiency of old-fashioned appliances and methods adopted to meet modern requirements. The working of my new plant will be effected without the aid of the expensive Bessemer blowing engines, such as required for the converters; a rough fining of the liquid pig iron, tapped directly into the compound steel furnace C by means of cold or hot blast from the blast-furnace blowing engines, may be obtained from a pipe connection f. Further, the expensive buildings necessary for the accommodation of the long rows of open-hearth furnaces of to-day can likewise all be dispensed with, together with the entire gas producer plants. The building D, as shown, would not take more space than that of the attached cast houses of a modern blast furnace plant.
The furnace C will cost about twice as much as a modern large stationary open-hearth and the same amount as an up-to-date "Talbot" or other big tilting furnace. E represents the apparatus for increasing the quality of the blast furnace gas to be used for heating purposes in the open-hearth. A pipe connection g for this purpose may branch off somewhere conveniently from the furnace gas main, and is attached at h to the reversing gas valve i of the furnace; k indicates the flue to any of the furnace smokestacks.
The steel furnace called "compound furnace" hereafter, on account of the combined open-hearth process and converter method, is shown in Fig. 2 and Fig. 3. Fig. 2 is a plan view; Fig. 3 a longitudinal section, and C in Fig. 1 is a cross-section.

The inclined air—blast nozzles, acting on or in the bath of the metal, are shown in C and in Figs. 2 and 3 as well. The "improving towers" for gas are E throughout; the gas regenerators, in this case better called "reheaters," are shown at G, and the reheaters for air at F.
As the usual furnaces are too well known to the readers of this journal I will not waste space and time in further description, but will shortly finish the main object, namely, the proof that blast furnace gas can be used in open-hearths, and that there is enough in surplus to run the steel plant when "compound steel" is manufactured.
I refer here to an invention of Mr. Ehrenwerth as long ago as 1883, which relates to the regeneration of blast furnace gas.
In von Ehrenwerth's calculations the absolute calorific value of blast furnace gas of poor quality is given as about 705 Calories per cubic meter, or about 80 B.T.U. per cubic foot.

Some time later other authors also made experiments, and found that blast furnace gas of about 932 Calories would give a suitable furnace heating gas of up to 1,472 Calories, or of a heating value of 160 B.T.U. per cubic foot, better than any standard gas producer gas. Since modern gas engines can also successfully be operated on the raw gas and since ore-roasting furnaces, lime-burning cupolas, etc., have also been successfully operated on this raw gas, therefore there is no doubt at all that even high heats as required in melting furnaces can be obtained from blast furnace gas.
In general, an open-hearth furnace of, say, 50 tons capacity, will use about 800 pounds of coal per ton of the product. The output per day is about 100 tons steel, which require, therefore, 80,000 pounds of coal to be gasified in the gas producers in 24 hours, or 3,333 pounds of coal per hour.
One pound of coal yields about 75 cubic feet of producer gas. Hence in our plant we have about 250,000 cubic feet of gas per hour, with an average heating value of approximately 130 B.T.U. One hundred tons of steel require, therefore, 80,000 X 75 X 130 = 780,000,000 B. T. U., or 1 ton, requires 7,800,000 B.T.U. for manufacture.
My intention is now to convert the 576 tons of pig iron in the same time in which they are produced into steel of open-hearth quality in a single furnace. If we assume a tap in each 4 hours, 96 tons are tapped six times in a day of 24 hours. Four hours are sufficient to transform this iron into steel, after a preliminary rough fining of the metal by means 01 the hot blast, besides a modern, say, Talbot continuous process, even if iron ore in lieu of scrap is to be used to a considerable extent. The latter method, however, will turn out profitably only with the high scrap prices of to-day. This all considered, the 576 tons pig iron will yield in a continuous process about 600 tons of steel in 24 hours, or 25 tons per hour. Each ton requires, as shown before, 7,800,000 B.T.U., so that 195,-000,000 B.T.U. per hour are required for 25 tons per hour.
Conclusion
As formerly shown in this article, there are about 173,280,000 B.T.U. for further use left per hour from the blast furnace plant, if we do not take into consideration the improvement of the gas by regeneration. The latter will practically amount to, say, only 20 per cent in heating value; the theoretical result gives 30 and 40 per cent. If we add 20 per cent to above 175,000,000 B.T.U., the available total from the blast furnace gas is increased to 210,000,000 B.T.U.
We have, therefore, a surplus of 15,000,000 B.T.U. over the required 195,000,000 B.T.U. per hour, so that the "regenerators" do not need to be continuously in operation. The coke required to perform this chemical improvement of the gas is easily obtained without cost from the waste of the coke-oven braize and the waste of small coke from the blast furnace plant.
The calculations prove, therefore, that this new process for manufacturing "compound steel" could be worked with blast furnace gas in a rational manner, since the expenditure in gaseous fuel practically falls considerably below that of the equivalent amount of solid fuel.
Any discussion of this subject and any criticism will be very much appreciated by the author of these lines, and any proof of an error in one of the facts or opinions given above will be noted with thanks in the interest of modern progressive work.